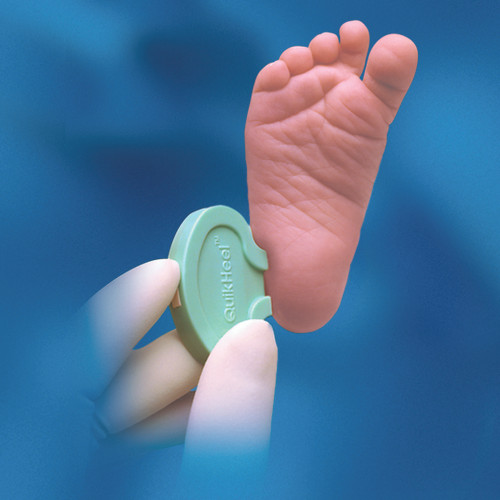
Description
- Designed to deliver precise incision during neonatal blood sampling
- Meets OSHA and NCCLS guidelines, featuring a permanently retractable blade and a shallow blade depth that meets infant safety standards
- Versatile grips fo ease of use
- Alignment guides for precise placement
- Color-coded body for easy size identification
- Quick-release sterility cap to prevent accidental incisions
- Extra Gentel Incision Device
| SKU # | 975533 |
| Manufacturer # | AT 1250 |
| Brand | Unistik Tiny Touch |
| Manufacturer | Owen Mumford |
| Country of Origin | China |
| Activation Method | Push Button Activation |
| Application | Pediatric Lancet |
| Blade Size | 2.5 mm Blade |
| Depth Settings | 1.0 mm Depth |
| FSA Eligible - Buy UOM | Yes |
| FSA Eligible - Primary UOM | Yes |
| FSA Eligible - Sell UOM | Yes |
| HCPCS | A4258 (Disclaimer) |
| Incision Type | Blade |
| Is_Active_Vendor | Y |
| Is_DSCSA | N |
| Is_Discontinued | N |
| Is_Medical_Device | N |
| Lot_Tracking_Flag | N |
| On_Allocation | N |
| Safety Feature | Retractable |
| Sterility | Sterile |
| Supplier_ID | 3883853 |
| Target Area | Heel |
| UNSPSC Code | 41104102 |
| Usage | Disposable |
Product Details
- Application:
- Pediatric Lancet
- Activation Method:
- Push Button Activation
- QTY Per Sell:
- 50
- Sterility:
- Sterile
- Usage:
- Disposable
- Depth Settings:
- 1.0 mm Depth
- Incision Type:
- Blade
- Safety Feature:
- Retractable
- Target Area:
- Heel
- Is New (Last 30):
- No
- Legend Code:
- No
- FSA Eligible:
- Yes
- Country of Origin:
- China